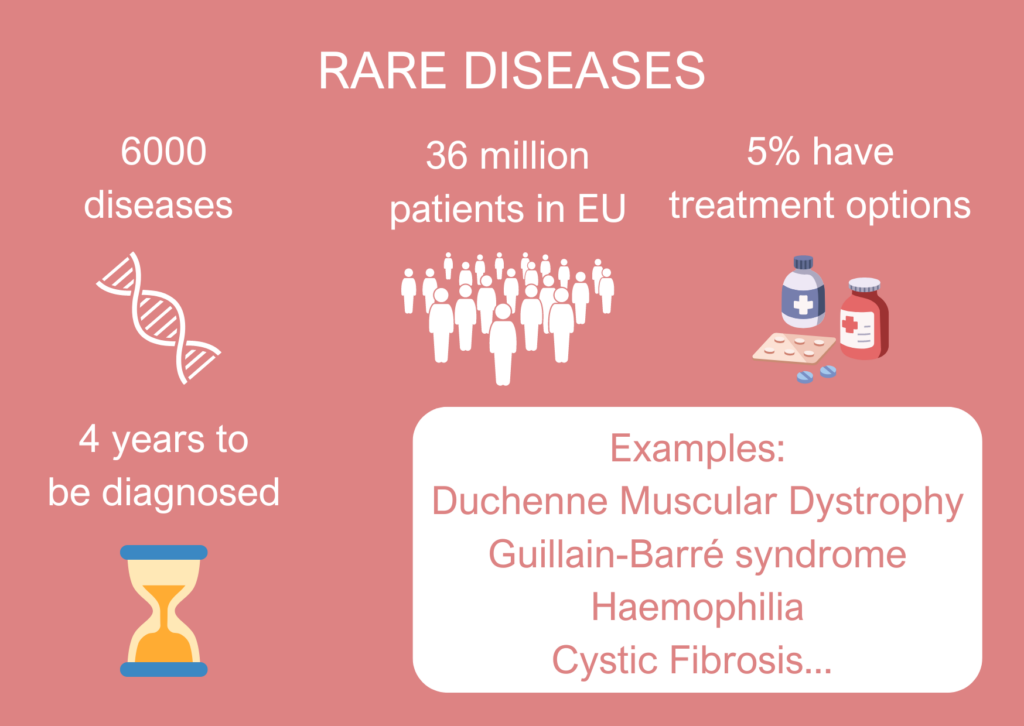
Rare diseases, defined as those affecting no more than one person in 2,000 are, in fact, not rare. It is estimated that 6,000 distinct rare diseases impact between 30 and 36 million people in the EU. However, the limited volume of patients for each disease hinders research, to the detriment of understanding of their biology, recruitment to clinical trials and survival rates. Therefore, pooling scarce resources spread across the EU is essential, underscoring the critical role the EU plays in this specific field.
What has the EU done for rare diseases?
When it comes to Rare Diseases (RD), the EU’s goal is to facilitate information and expertise sharing between patients and professionals all over the Union.
Since 2018, issues related to RD have been addressed at the European Commission level, by the Steering Group on Health Promotion, Disease Prevention and Management of Non-Communicable Diseases which has been replaced in 2022 by the Expert Group on Public Health.
It must be noted that most of the EU’s actions regarding RD aim to boost research. The ERICA project, which runs until February 2025 and benefits from a €2 million Horizon 2020 grant, intends to coordinate the clinical research activities of the European Reference Networks (ERNs). ERICA unites the expertise of the 24 ERNs and creates a collaborative platform to share knowledge and good practices through the assembly of transdisciplinary research groups across the different medical areas.
The EU has supported the coordination between research funders across Europe and beyond, by launching in 2019 the European Joint Programme on Rare Diseases (EJP RD) which gathers research funders, universities, research organisations and infrastructures, hospitals and patient organisations from more than 135 institutions in 35 countries (26 in the EU). The EJPRD will launch a new European Partnership on rare diseases called the European Rare Diseases Research Alliance – ERDERA co-funded by Member States and the Commission under Horizon Europe (EU funding up to €150 million) in the fall 2024. ERDERA aims to develop new diagnostic methods and pathways, support concrete health benefits to RD patients, through better prevention, diagnosis, and treatment and to ensure strong alignment of strategies in rare diseases research across countries and regions.
Moreover, the EU is supporting the definition, codification and inventory of rare diseases by creating Orphanet, the portal for rare diseases and orphan drugs. Orphanet aims to provide high-quality information on rare diseases and ensure equal access to knowledge for all stakeholders. Orphanet also maintains the Orphanet rare disease nomenclature (ORPHAcode), essential in improving the visibility of rare diseases in health and research information systems. Orphanet is therefore involved in many EU-funded research projects on rare diseases (e.g. ERICA, EJP RD etc.).
What is the impact of other EU legislations on rare diseases?
Many existing or upcoming EU health legislations have impacts on RD. For example, the European Health Data Space (EHDS) will provide a consistent, trustworthy, and efficient system for reusing health data for research, innovation, policy-making, and regulatory activities (secondary use of data) The EHDS specifically mentions RD in several articles, particularly supporting diagnosis and treatment. It would foster RD research by providing an avenue forward for some parts of the data sharing necessary to create a multi-country integrated rare disease ecosystem.
In 2023, the Commission put forward a ‘pharmaceutical package‘ to revise the EU’s pharmaceutical legislation. The package includes proposals for a new directive and a new regulation, which replace the existing pharmaceutical legislation, including the legislation on medicines for children and for rare diseases. The Parliament adopted its position in April 2024, stating that medicines developed to treat rare diseases (‘orphan drugs’) would benefit from up to 11 years of ‘market exclusivity’ (protecting them from competition from similar medicines with similar indications, which could not be marketed during the exclusivity period) if they met a ‘high unmet medical need’. The file is now in the hands of the Council, which has not yet adopted its position and work is ongoing under the Hungarian Presidency.
And now what will happen to rare diseases EU policies?
Last June, EU citizens elected their MEPs who will sit in the European Parliament from 2024 until 2029. The new MEPs will gather for the opening plenary in Strasbourg from 16 to 19 July where they will decide on the makeup of its standing and sub-committees. In 2023 a new sub-committee (with no legislative power) on public health was created. Its future is uncertain but many hope that it will become a fully-fledged committee of the European Parliament. Health issues in Parliament are currently dealt with in the Committee on the Environment, Public Health and Food Safety (ENVI). Having a standalone public health committee would give greater visibility and importance to health issues in the Parliament. This is supported by the EPP group the largest political group in the Parliament but opposed by S&D, the second largest group.
Subsequently, MEPs will elect the new President of the European Commission and, in the fall, approve or reject the nominees 26 commissioners for the next five years. We do not know yet who the candidate for EU Commissioner for Health and Food Safety will be.
In February 2024, Europe’s health budget is being cut by €1 billion to help finance the €50 billion support package to Ukraine. This worrying 20% reduction of the EU4Health Programme’s budget alarmed civil society and patient groups, especially as many predict the health budget will not return to pre-COVID levels. EURORDIS-Rare Diseases Europe, a non-profit alliance of over 1,000 rare disease patient organisations urged new MEPs to keep health at the forefront. This cut was the doing of Hungarian Prime Minister Viktor Orbán who refused to increase the overall EU budget to provide support to Ukraine.
Speaking of Hungary, the country is set to play a significant role in the next six months. Hungary is taking over from Belgium as the Presidency of the Council from 1st July to 31st December 2024. In terms of health, the Hungarian Presidency will prioritise cardiovascular diseases and organ transplantation. Regarding rare diseases, its programme merely states, “we intend to allow room for discussion on other topics such as rare diseases, the links between mental health and various emergencies, and the possibilities for cooperation between Member States on the affordability of medicines.”
On 21st of June the Employment, Social Policy, Health and Consumer Affairs Council adopted its Conclusions on the European Health Union which encourage to continue and strengthen the work on rare diseases among other areas. Malta was one of the few countries that expressed some criticism over these conclusions: Health Minister Jo Etienne Abela said Malta would have liked to see “a more concrete roadmap as to how Member States can work together in the interest of patients.” He also said Malta would favour joint procurement for orphan drugs. This mechanism allows Member States and other European countries to procure medical countermeasures collectively. It was crucial in the collective purchase of vaccines and personal protective equipment during the COVID-19 pandemic.
All these developments regarding health policy have prompted EURORDIS to call for a European Action Plan on Rare Diseases to drive a common strategy across Member States. Such an action plan can put the spotlight on rare diseases as a public health priority and provide solutions to the unmet needs of these 36 million people.
If your company or organisation is involved in rare diseases and would like to raise the bar in the EU on this crucial topic for millions of citizens, please do not hesitate to contact us. Lykke Advice can help you make your voice heard by EU policymakers and navigate EU policies and programmes, contributing to a healthier future for all, ensuring that no patients are left behind.


