Life sciences in a snapshot
Two weeks ago, the Commission released its Life Sciences Strategy named “Choose Europe for life sciences” a title reminiscent of the “Choose Europe for Science” initiative of Emmanuel Macron to attract American researchers to the Old Continent.
Life sciences are defined in the Strategy as “the study of living systems, from human beings, animals, plants, microorganisms to ecosystems and their interconnectedness, via a number of often interrelated disciplines”. Life sciences have applications in several sectors such as health, food, agriculture and sustainability.
The life sciences sector is a crucial pillar of the European Union’s economy, accounting for 13.6% of total employment and contributing 9.4% to the EU’s GDP. Primary production, food, and the bio-based manufacturing sector (which includes agriculture, forestry, fishing, food and beverage production, and other related industries) are the main areas for life sciences, supporting 8 million jobs and generating EUR 785 billion in value. In comparison, human health technologies and services account for 9.5 million jobs and EUR 622 billion. These figures underscore the significance of the life sciences sector not only for advancing innovation and improving health outcomes, but also as a key engine of economic growth and employment across EU Member States. The sector holds particular strategic and economic weight in countries such as Germany, France, Denmark, Ireland, and Belgium.
A closer look at the EU Strategy
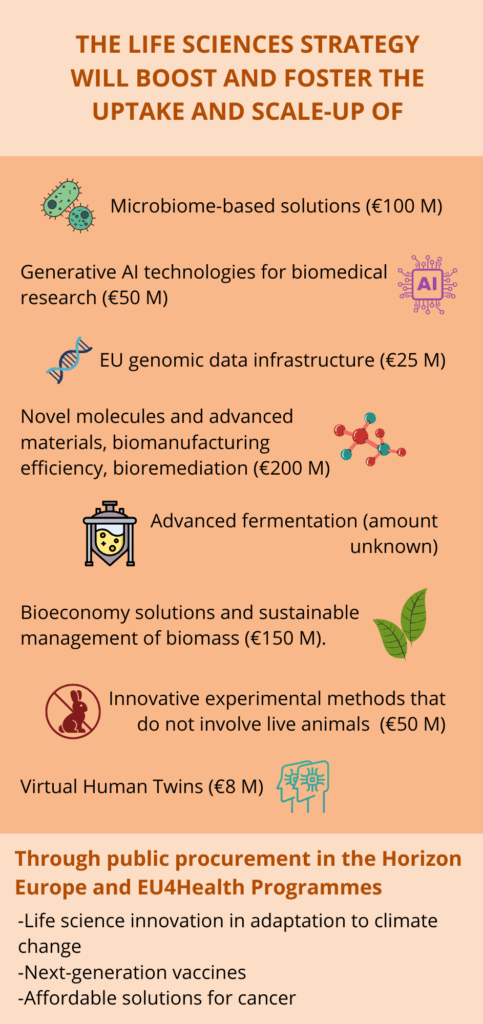
The Strategy places a strong emphasis on research and development (R&D) as well as on research and innovation (R&I). While this focus is commendable, it somewhat overlooks the fact that the EU is already performing well in these areas with world-class research institutions and infrastructures. As Europe struggles to put its innovations in the market as expressed in the Draghi report, there is more urgency to help innovative life sciences solutions grow beyond the lab and reach commercial viability. However, some specific applications or technologies are named in the document and will receive targeted support as shown in the infographics below.
Moreover, the strategy appears to prioritise the health-related dimensions of life sciences, with less attention given to other aspects such as food systems, agriculture, and environmental sustainability. While health is a key sector, this narrow focus may limit life sciences’ contribution to broader societal and environmental challenges. Nevertheless, attention to clinical trials, crucial to testing new treatments and medical devices, is a welcome development. Indeed, the pharma industry reported that Europe is falling behind China and the U.S when it comes to the number of clinical trials performed and number of patients enrolled. The Commission said it will closely monitor the implementation of the Clinical Trials Regulation adopted in 2022. It will also propose an investment plan to facilitate multi-country clinical trials. Such trials are generally more robust than single-country ones because they enable faster patient recruitment, increase population diversity, and enhance the global applicability of results, which is especially important for rare diseases, for instance.
In addition, the Strategy has some provisions on Science, Technology, Engineering, and Mathematics (STEM) education and skills which are key to the sector’s competitiveness and growth. The Commission will support the career development of life science researchers and help researchers from non-EU countries set up in the EU, notably through the Choose Europe initiative. The Commission will also launch a study in 2025 to identify the competences, skills and training needs for the life sciences sector. This study is a good first step but it will be important that its results are followed by concrete actions financed by earmarked budgets.
Making the most of the Strategy
The Life Sciences Strategy is interconnected with upcoming legislations and initiatives such as the Biotech Act, the revised Bioeconomy Strategy, the revision of the Medical Devices and In Vitro Diagnostics Regulations, the EU Climate Adaptation Plan, the Strategy for AI in Science and the AI Factories initiative, as well as the Advanced Materials Act. Ensuring coherence and strategic alignment among these frameworks will be essential to prevent regulatory overlaps. Even if most of these files are expected for the end of 2025 or 2026, stakeholders wishing to influence these legislations must act now when Commission officials will be drafting the different texts.
Overall, the Strategy will be supported by an annual investment of EUR 10 billion. This is financed mostly by the Horizon Europe Programme but also by the Interregional Innovation Investment, the upcoming Scaleup Europe Fund and the Digital Europe Programme. The Commission also announced it will de-risk the development of new medicinal products and medical devices through a new European Research Area policy action. Given the wide scope and diversity of these funding instruments, SMEs active in the life sciences sector should closely monitor the publication of relevant calls under each of these programmes to seize timely opportunities for support in R&D, scaling up, cross-regional collaboration, and digital transformation. Active engagement with the EU funding landscape will be essential for these companies to ensure they can fully benefit from the EU’s strategic investment in the sector.
The Strategy also foresees the establishment of various groups, platforms, and tools that will be strategically important for SMEs in the life sciences sector to engage with and leverage to boost their visibility and defend their specific interests. The Life Science Coordination Group, bringing together stakeholders from industry, academia and civil society, will be set up in 2025 to ensure innovation friendly coherent policies, funding and activities and to monitor progress in implementing the Strategy.
The group will also create an AI-powered interactive tool to help researchers and innovators navigate the EU regulatory landscape, particularly in the early stages of research and development. While such a tool is a valuable starting point, navigating regulatory complexity, tailoring strategic positioning, and securing the right partnerships still requires expertise—making the support of a specialised consultancy such as Lykke Advice indispensable.
Additionally, to help life science startups in their journey to the market, the Commission will launch in 2026 a matchmaking strategic interface connecting life science startups, industry and investors. The Life Sciences Investors & Corporates Interface will be a key platform for startups seeking funding.
Together, these mechanisms offer SMEs both an opportunity to have a voice in policymaking and practical support for innovation and market entry. Lykke Advice is a boutique consultancy specializing in helping SMEs use public affairs to open new markets in the EU, please do not hesitate to contact us if you wish to seize the opportunities brought by the Life Sciences Strategy.


